%ABV = (OG – FG) x 131.2
You have seen the above formula in numerous online posts and books. I even include it in my book. But, is it the best formula for estimating the percentage of Alcohol by Volume (%ABV) in hard cider? What does it even mean? I started this post originally planning to write it up as a Mālus Trivium but soon realized I really needed to spend more time on it. The equation is fairly straight forward. You take your original gravity (OG) and subtract your final gravity (FG). This difference is multiplied by 131.2 and the result is your estimated %ABV. But, if you are like me, you might have a few questions.
- What does the OG and FG represent?
- Why 131.2? Is it the right number?
- Why is this only an estimate? Can’t we be exact?
While I will talk about accuracy later, I always try to write about the science of cider in a way to help everyone understand. Sometimes, I am sure that I fail but I will keep trying. To make this post clearer, I think it’s easiest to assess this question under the assumption that mass is conserved in the fermentation process. What that means is that the mass of sugar that is fermented is converted into ethanol and CO2 of the same total mass. If you start out with 100 grams of sugar in your apple juice, this sugar is converted into 100 grams of ethanol and CO2. We will discuss this in more detail but the assumption of conservation of mass for the chemical process of fermentation helps explain the common %ABV equation.
What does OG and FG represent?
The Original Gravity (OG) represents the amount of sugar in the juice before the fermentation. The Final Gravity (FG) represents the amount of sugar, if any, remaining in the hard cider after fermentation. Remember that I said for this equation we are assuming mass is conserved. If you have 100 grams of sugar, you should produce ethanol and CO2 equal to 100 grams. However, the difference between the OG and the FG means something happened. You lost mass during the fermentation process. Remember all those bubbles going through your airlock? That was CO2 escaping and that is the reason your OG and FG are different. The mass of CO2 escaped and reduced your specific gravity. CO2 has mass and fermentation generates a lot of it.
Let’s assume your OG was 1.050 and your FG is 1.000. This means that your cider lost 0.050 grams of mass per ml of cider. We will assume all the CO2 generated escaped through the airlock. Therefore, we know we lost 0.050 grams of CO2 per ml of cider. We also know that since mass is conserved, we created 1.05 grams of ethanol for every gram of CO2 that was created and lost. We know this because one glucose molecule (180 grams) is converted into two ethanol molecules (46 grams each) and two carbon dioxide molecules (44 grams each). Therefore, The 180 grams of glucose ferments into 92 grams of ethanol and 88 grams of CO2. The 180 gram mass of glucose is conserved. The ratio of ethanol, 92, to CO2, 88, is 1.05, which means every gram of CO2 generates 1.05 grams of ethanol.
Glucose to Ethanol and Carbon Dioxide Reaction: Molar Mass is Conserved
C6H12O6 -> 2C2H6O + 2CO2
In my above example with a 1.050 OG, our specific gravity readings tell us we lost 0.05 grams of CO2 per ml of cider during fermentation. That means we also generated 0.0525 grams of ethanol per ml of cider. Knowing the mass of ethanol generated allows us to divide this amount by the density of ethanol, 0.8 grams per ml, and multiple it by 100. This gives us the percentage of ethanol per volume of cider, which is 6.56% (0.0525/.08*100). A change in specific gravity of 0.05 means, 0.0525 grams of ethanol were created. That’s means your alcohol by volume of your hard cider would be 6.56%. Wait, I never mentioned the 131.2 number.
Why the 131.2 number?
The quick answer is the ratio of the grams of ethanol to grams of CO2, 1.05, divided by the density of ethanol, 0.80, times 100 to represent the results as a percentage equals 131.2. The long answer is the 131.2 probably isn’t the best number. For example, the 1.05 is really 1.0468 and the density of ethanol at 20C is 0.78923 and not 0.8. This would make the factor 132.7, which would increase the estimated %ABV from 6.56% to 6.64%. This starts to highlight why the %ABV is just an estimate. It also helps identify what drives the variation and why this is just an estimate. This variation is not in the 100, which is just used to represent the number as a percentage. That means our 131.2 becomes 1.312. Also, the density of ethanol is well known. It changes with temperature because density includes volume and volume changes with temperature. We can consider the density a constant so the 0.8 or 0.78923 factor means the number in question is the 1.05 or 1.0468. Let’s explore this number and why it makes this calculation an estimate.
Why is this an estimate?
There are two main areas of uncertainty in the fermentation process. One is the efficiency of the fermentation process and the other is the amount and types of sugar in your juice. Louis Pasteur is often cited for his work on fermentation. He proved that fermentation was not a decomposition process but actually driven by an organism, yeast, which he publish in papers in 1857-1858. As noted in Claude Jolicoeur’s book (1), Pasteur identified that 100 grams of sugar are converted into 48.4 grams of ethanol, 46.6 grams of CO2, 3.2 grams of glycerin, 0.6 grams of succinic acid, and 1.2 grams of yeast cells. This has been refined over additional research but it highlights that it’s not only ethanol and CO2 being produced. We also know that the yeast strains influence this process. Mutating yeast by eliminating or adding genes, can impact how efficient yeast are in the fermentation process. In addition, we know that sugar is consumed both by aerobic and anaerobic processes with the yeast cells. Usually, the aerobic process is during the reproductive stage and the anaerobic is for the production of alcohol. Now, imagine the complex microflora and how it uses sugar and other solids to create various chemical reactions. Maybe the 1.05 should be 1.02 or 1.06 depending on the microflora. There is a lot going on in the fermentation of your hard cider and this isn’t even the biggest variable, which is the sugar.
The standard %ABV calculation makes two broad assumptions about the sugar in your juice and cider. The first is that the sugar is all a simple reducing sugar like glucose. We know apples contain glucose, fructose, and sucrose. We also know that fructose is the most dominate sugar but it has the same elements, C6H12O6, as glucose. Sucrose, on the other hand, breaks down into one glucose molecule and one fructose molecule so ultimately, all of these sugars should break down nicely into ethanol and CO2, just like glucose. However, most saccharomyces yeast prefer glucose over fructose. Also, sucrose requires an invertase process to break it down into glucose and fructose. This highlights another reason how there are slight variation in fermentation caused by the DNA of the yeast being used. The amazing process of fermentation can also include the fermentation of some supposedly non-fermentable compounds like sorbitol. There is a process, though not common in most hard cider fermentation, that can convert sorbitol into fructose, which will ferment into ethanol.
The second and biggest impact is the assumption that all the suspended solids are sugar and not other things like acids, phenols, pectin, sugar alcohols, and other compounds that make our hard cider taste the way it does. When you use your hydrometer or refractometer to measure specific gravity or Brix, the measurement includes all these dissolved solids. You may think that because your hydrometer is reading 1.000 it proves there really aren’t any solids or they are so small that they don’t measure. However, don’t forget the impact of ethanol, which has a much lower specific gravity, 0.78923, than water. A rough calculation says that 6% ethanol in pure water should make the mixture have a density near 0.987.
The dissolved solids explain why you would have specific gravity measurements at or above 1.000 on your hydrometer and not have any actual sugar remaining. This is why it’s also hard to judge whether you actually have a lot or a little sugar left if you are trying to decide when to bottle for natural carbonation. In his book, Jolicoeur examined hundreds of apple samples from around the world and found that the non-sugar solids represent about 18% of the total dissolved solids. If your hydrometer says your juice has 100 grams per liter (~1.040), only 82 grams of that is probably fermentable sugar. Instead of your juice measuring 1.040, it would be like it was 1.033. That means your %ABV would really only be 4.33% instead of 5.25% using the standard equation.
What does this all mean?
What this means is that we continue to need more research into hard cider and the process for making hard cider. It’s a great reminder to support your universities and professionals dedicated to learning and sharing what they find. It also means that we need to continue to challenge the concept that everything we know from beer or wine applies directly to cider. Some of it does but some of it doesn’t or does but not fully. In the case of estimating your %ABV, we should probably follow Claude Jolicoeur’s advice and use a factor closer to 128 versus 131.2 for our calculation(1). It’s not a huge difference, a specific gravity of 1.040 would have a potential %ABV of 5.12 versus 5.25, but it better reflects our industry , which is something we should all embrace.
Estimating %ABV: The Best Equation for Cider
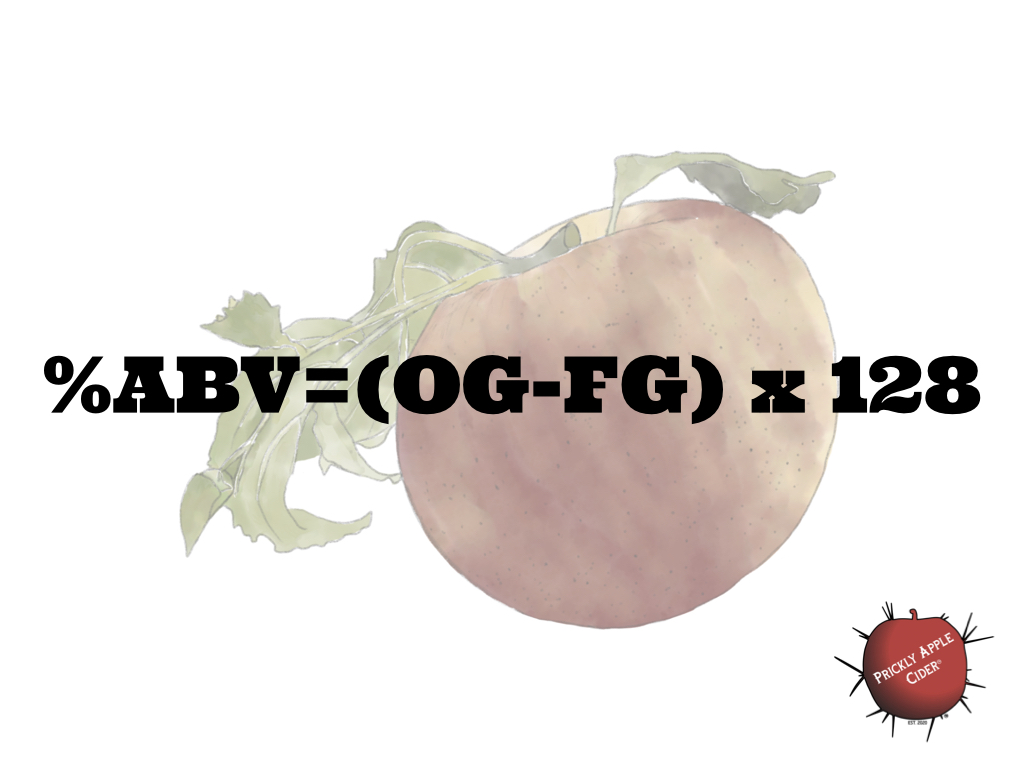
(1) Claude Jolicoeur, The New Cider Maker’s Handbook, 2013.
Don’t miss any future Mālus Trivium articles. Follow me and you will get a link to my latest article delivered to your inbox. It’s that easy!

















