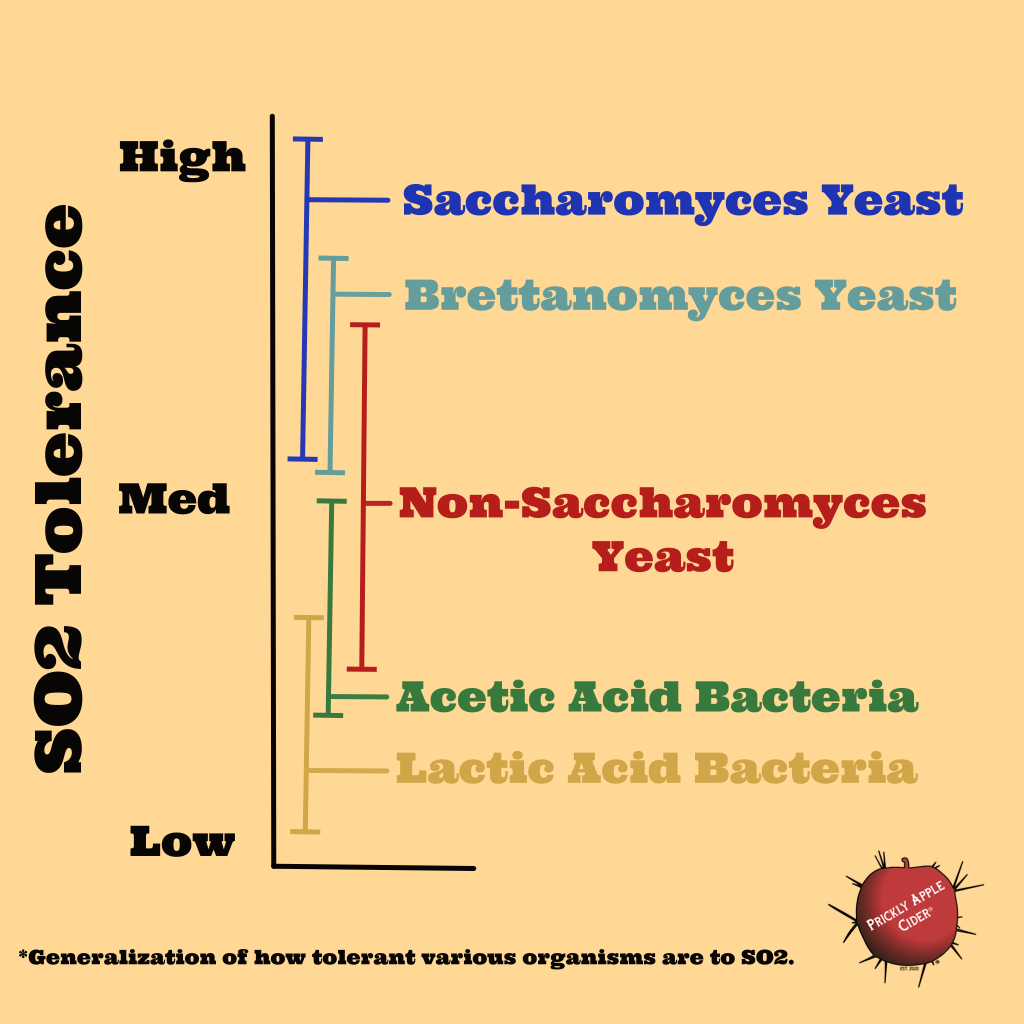
Sulfur dioxide (SO2) has the ability to weaken the cell walls of organisms resulting in their death or inability to function. It can also react and bind with phenolic and other compounds impacting color, sweetness, bitterness, and astringency. SO2 tolerance is the ability of an organism to resist being impacted by exposure to SO2. Sulfur dioxide is often called sulfite or sulphite depending on the version of English you speak. This is accomplished by adding potassium or sodium metabisulfite to your juice or cider. This is often called by the brand name of Campden. When you add the metabisulfite, it forms sulfite or SO2. The sulfite binds with organisms and compounds and are often referred to as bound. Bound sulfites are no longer active. Only the free sulfites or what is often called SO2 is active and available to bind with organisms and compounds. Free SO2 is generally measured in parts per million (ppm) or mg/liter. They are approximately equivalent so 10ppm is the same as 10mg/l. I know someone will ask, what ppm do the low, medium, and high levels represent in my graphic. As I often say, the answer is that it depends. I can give you a rough idea of high as most research doesn’t go above 200ppm (~200mg/l) of free SO2 and many regulations limit the amount of sulfites you can add, which is often around 100ppm. But the challenge is that there are so many different strains and organisms within each of the main organisms I listed that you can’t really put a clear number on when something is impacted. Also, the pH of the cider matters because acids help break down the cell walls too. The graphic is a generalization to highlight the usually hierarchical nature of how organisms are impacted by SO2.
Saccharomyces yeast is the most common yeast used for wine, beer, bread, and most fermented foods and beverages for a reason. Besides being a strong fermenter, it is generally very tolerant of SO2. In fact, yeast are generally more tolerant of SO2 than bacteria. When you apply sulfites to your hard cider, the first thing you are eliminating is bacteria and generally, the lactic acid bacteria (LAB). LAB is the organism that performs malolactic fermentation (MLF), which is a process where malic acid is converted into less harsh lactic acid. If you are using culinary apples to make your hard cider, MLF is potentially something you want as your cider may be dry, acidic, and lacking any tannins to provide balance. Remember that the effectiveness of SO2 is pH dependent. As your pH drops, SO2 become much more effective so even a small amount can start inhibiting even high SO2 tolerant organisms. After SO2 bind and effectively start inhibiting bacteria, it also starts to work on many weaker fermenting yeast or what is commonly found on the apple and pear fruit. If you are planning a natural or wild ferment, these yeasts are generally the next organisms that start to be inhibited. Yeasts like Candida, Lachancea, and Pichia are generally more susceptible to SO2, though not always. Brettanomyces also has a range of tolerance depending on the strain. One interesting aspect showing in many research papers is that these organisms appear to be evolving and they are becoming more SO2 tolerant. It is not uncommon to find Brettanomyces strains capable of surviving at very high levels of SO2. This isn’t surprising. Look at Saccharomyces cerevisiae yeast strains. While these can be found on fruit and plants, those are not common places and it is hypothesized that it is actually a yeast that has evolved around human habitation. I have yet to find any research that identifies the origin of Saccharomyces yeast. One hypothesis is that through thousands of years of cohabitation, the yeast has evolved to perform best in the environments most commonly made for it by humans.
As always, I hope this session of Mālus Trivium has gotten you thinking more about how you make your cider and how things are always connected.
Did you enjoy this article? Don’t miss future posts from PricklyCider.com by following us today! PricklyCider.com is your source for all things cider.




